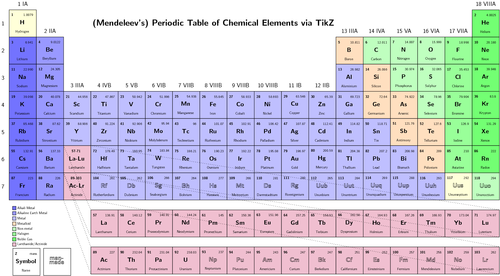
Subject: Science, Chemistry lesson on the Periodic Table.
Lesson time: 2 class periods 50 minutes per class
(ASSURE model.)
A= Analyze your Learners
Suitable for grade 9,10,11,12. Ages 13 and up. Both males and females with a diverse background.
Information needed to know about these learners:
- What science classes have students had previously?
- Are they ELL or do they have an IEP?
- What are their cultural or demographic backgrounds?
- What skill level does the students have regarding technology use?
- What is their academic background?
S = State your Objectives
Objectives:
Objective 1: Given a diagram of the periodic table, the students will be able to identify and differentiate between groups and periods.
Objective 2: Students should be able to categorize elements based the relationship between periods and energy levels for the first 20 elements.
Objective 3: Students should be able to identify the electron configuration for a given elements based on its location on the periodic table.
S = Select Media and Materials
A large periodic table is displayed in the front of the classroom. The teacher will demonstrate the organization of the periodic table using the wall poster.
Computers with basic office applications installed as well as internet access.
- Students will view the following you tube video to understand the periodic table of elements: https://www.youtube.com/watch?v=bnavG6iXUb0
- Students will access the build an atom site at http://phet.colorado.edu/en/simulation/build-an-atom to review the structure of atoms and how they are organized by protons and outer electrons
- Students will create a power point presentation of the first 20 elements including the name, symbol, number of protons, neutrons and electrons. The number of electrons in the outer shell (valence electrons) and the Lewis dot structure will also be included. The students should include which atoms are grouped together by valence electrons.
Students will begin this project on the first day and finish on the second day. Students can work in groups of four. Each student can research 5 elements. The power point should show continuity. Each element should have one slide. There should be a title slide and a reference slide. The title slide should include the names of all participants in your group.
Slides should be creative. When each student has finished their five elements the groups can combine their work into one presentation. Each group can then present a portion of their elements until all the elements have been reviewed.
U = Utilize Media and Material
- Teacher will first prepare the technology center computers with the appropriate links to the sites needed for the lesson. If needed, reservation for use of the technology center must be made.
- Teacher must ensure the computers are in working order, that there is internet access, that the proper software has been installed and the correct web sites have been bookmarked for students to find quickly and that the links still work.
- Students will utilize the technology center to gain access to the internet to review the video and build an atom site. Students will then utilize the power point software to create their presentation.
R = Require Learner Participation (Knowledge, Comprehension, Application, Analysis)
- Students will utilize the "Build an atom" site to review the structure of the atom. This review will reinforce the relationship of the structure of the atom to the periodic table with regard to protons and electrons.
- Students will then log on to computers and click the preset link under the class tab for the video to understand and review the organization of the periodic table.
- Students will create flashcards for required terms with definitions using http://www.kitzkikz.com/flashcards/
- Students will utilize their knowledge of the structure of atoms to create a power point presentation of the first 20 elements. They will utilize the software to create a presentation including the information stated in the rubric for each element. The students will share thier presentations with the class and post them to the class web site.
Day 1:
Student activities. Students will work on the following computer based activities in groups or alone. The teacher will walk around and monitor student progress.
A large periodic table is displayed in the front of the classroom. Students will research the definitions of the following terms. They will produce flash cards using http://www.kitzkikz.com/flashcards/ 20 minutes.
Atom – simplest form of matter
Element - A substance that cannot be broken down into simpler substances by chemical means. An element is composed of atoms that have the same atomic number, that is, each atom has the same number of protons in its nucleus as all other atoms of that element.
Proton – positive charged particle, found in nucleus of atom, has mass
Neutron – no charge particle, found in nucleus of atom, has mass
Electron – Negative charged particle, found in orbitals around nucleus, has very little mass
Atomic number – the number of protons found in an element
Atomic mass – the number of protons and neutrons found in element
Group – columns on the periodic table, contain like elements
Row – also called periods on periodic table
Review of periodic table and structure of atoms. 15 Minutes
Students will view the following you tube video to understand the periodic table of elements: https://www.youtube.com/watch?v=bnavG6iXUb0
Students will access the build an atom site at http://phet.colorado.edu/en/simulation/build-an-atom
to review the structure of atoms and how they are organized by protons and outer electrons
Student assignment:
Students will create a power point presentation of the first 20 elements including the name, symbol, number of protons, neutrons and electrons. The number of electrons in the outer shell (valence electrons) and the Lewis dot structure or electron configuration will also be included. The students should include which atoms are grouped together by valence electrons.
Student Directions: 15 Minutes
1. Read the following directions carefully.
2. Your group of 4 students you will create a power point presentation of each of the first 20 elements on the periodic table. Assign 5 elements per person. The design of the presentation should be consistent throughout.
3. Each slide should include information on the element name, symbol, atomic number, atomic mass, number of protons, neutrons, electrons, electron configuration, the group number and row number. All information about each element should be presented on one slide. Be creative!
4. You should also include a title slide and a reference slide. There should be at least 22 slides in your group’s presentation.
5. A good resource for finding the information is your text book or http://www.endmemo.com/chem/
Example of the basic information on a power point slide of elements:
Hydrogen
Symbol – H
Atomic Number 1
Atomic Mass 1.008
Protons 1
Neutrons 1
Electrons 1
Electron configuration 1s1
Group 1 Row 1
Day 2: (Synthesis)
Complete the power point in groups for 30 - 40 minutes. During the last minutes of class each group will present a set of five slides. If there are more than four groups the number of slides can be adjusted.
If finished early or you do not wish to do presentations the students can complete the worksheets under the student worksheets tab and take the practice quiz on the practice quiz tab.
E = Evaluate and Revise
- Student learning can be assessed by their power point presentation and how they grouped the elements according to the rubric.
- Students will be given a worksheet with a table containing elements and spaces for number of electrons in the outer shell and the number of protons. The student will then fill in the valence electrons and the number of protons by looking up the element on the periodic table.
Revisions:
After lessons add any suggestions or comments
Rubric
Your browser does not support viewing this document. Click here to download the document.
